Right then I think this sections the most appropriate place for a guide.
If you have any old rusty bits or even bits and bobs you'd like to clean up and paint you can use electrolysis to clean up parts. It is very effective and requires very little physical effort. IT IS ALSO CHEAP.
What you need
A bucket or container (5 litre tub will do for most objects)
A 12v battery or battery charger
2 anodes (2 pieces of conductive metal)
Tap water (hot water works best)
Copper wire
Length of copper tube (or other conductive metal)
Electrolyte chemical
Essentially you'll be looking to setup the above equipment like so
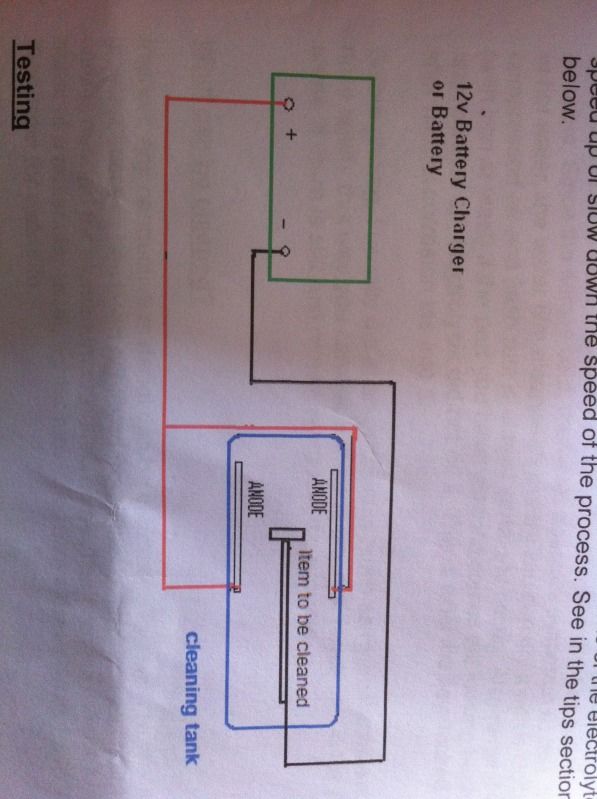
Step 1
Hang your anodes over the side of your container using the copper wire. Try not to submerge the copper wire if you can help it. Ensure that they are linked together. On mine I attached a crocodile clip at the end of the wire for an effective link with the power supply.
Provided that you keep the anodes clean they can be used over and over
Step 2 - mixing the electrolyte solution.
There's 2 different chemicals that you can use which are widely available and cheap.
Sodium carbonate - can be used on all metals and is kindest for any aluminium or precious metals. Is very good for removing rusts, paint, oil and general road grease. If you're using a 5 litre tub then just 25grams of sodium carbonate is sufficient combined with 4.5 litres of warm water.
Ammonium chloride - only to be used with steel and cast iron, In comparison it is quite savage and is much quicker and more aggressive than sodium carbonate. It isn't as good for degreasing. When mixing 35grams per 4.5 litres of warm water.
Several things to remember;
Wear goggles just incase
Stir thoroughly and always apply the chemical into the water rather than the other way round (not nice in eyes if the water splashes into your eyes if you've not worn goggles.
The solution will get hotter when the current is put through it
Personal experience - go with the sodium carbonate unless the item is very heavily corroded

Step 3 - connecting to your power supply
Positive terminal/lead should connect to your anodes
Negative terminal/lead connects to your metal rod which should span the diameter of your tub. Use copper wire to hang the object that you need cleaning off the rod

Obviously don't let your rod physically touch any of the copper wire linking with your anodes else it'll spark or blow a fuse in the charger.
And that's about it. Remember to wear gloves if you can, marigolds are fine.
Always setup in a well ventilated area
When the lower supply is turned on there will start to be bubbles rising off the item and you'll start to see deposits seperating off the item being cleaned
The process generally doesn't take long, half an hour may suffice for a mildly dirty item. You can hang several items at once into the tub/bucket.
Rinse off then under a tap, I've bought some small wire brushes to aid in the process. If all the rust and deposits have gone you can then paint the item if you wish or even zinc plate it (that's another how to guide) the freshly cleaned item should be dried thoroughly and ideally primered or even sprayed with wd40 to prevent 'flash rusting' appearing.
You can reuse the electrolyte solution if you wish, just filter out the deposits which will eventually sink to the bottom.
You can use a small sieve and use that to clean multiple nuts and bolts. Just connect the negative supply to the sieve rather than use the rod & copper wire
You can obtain all of the parts here for well under £10. onviously more if you dont yet have a battery or battery charger.
And a couple more pics of the process & some of my results so far
Large amounts of rusty/greasy deposits form on the surface

Here's how well some bolts clean up. The bolt I the middle is an old torsion bar bolt which looked exactly the same as the one on the left before cleaning. You can even see the numbers stamped in the head again


